
The mole is used for this purpose.Ī mole is defined as the amount of a substance that contains the number of carbon atoms in exactly 12 g of isotopically pure carbon-12.
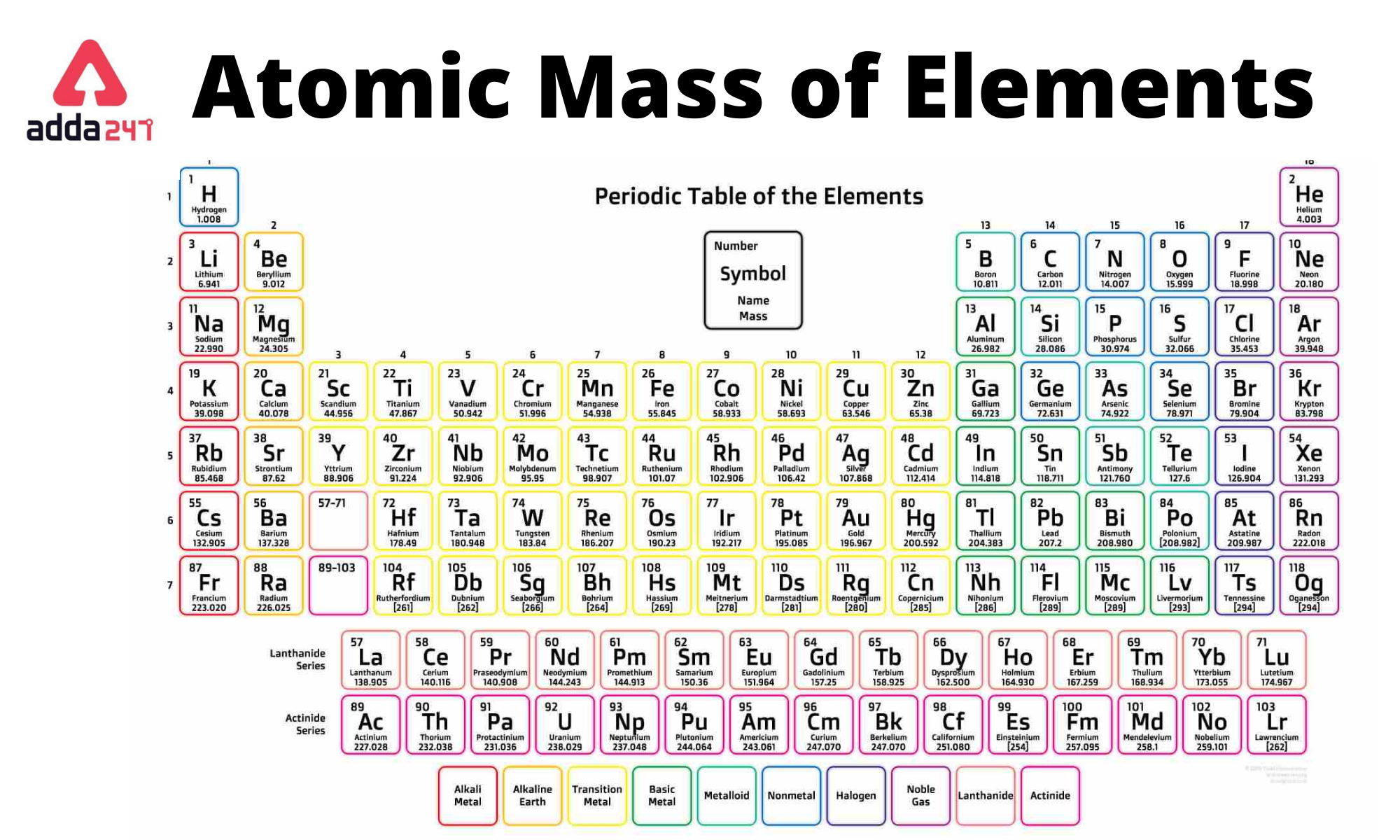
Any readily measurable mass of an element or compound contains an extraordinarily large number of atoms, molecules, or ions, so an extraordinarily large numerical unit is needed to count them. Atoms are so small, however, that even 500 atoms are too small to see or measure by most common techniques. Sheets of printer paper are packaged in reams of 500, a seemingly large number. For example, cans of soda come in a six-pack, eggs are sold by the dozen (12), and pencils often come in a gross (12 dozen, or 144). Many familiar items are sold in numerical quantities that have unusual names. The quantity of a substance that contains the same number of units (e.g., atoms or molecules) as the number of carbon atoms in exactly 12 g of isotopically pure carbon-12., from the Latin moles, meaning “pile” or “heap” ( not from the small subterranean animal!). The unit that provides this link is the mole (mol). To analyze the transformations that occur between individual atoms or molecules in a chemical reaction it is therefore absolutely essential for chemists to know how many atoms or molecules are contained in a measurable quantity in the laboratory-a given mass of sample.
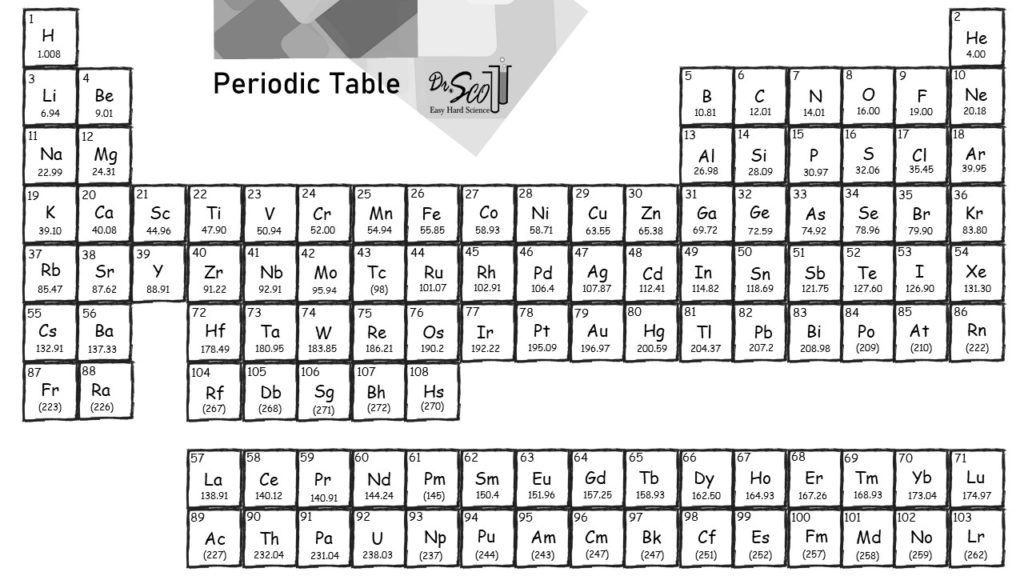
In the laboratory, for example, the masses of compounds and elements used by chemists typically range from milligrams to grams, while in industry, chemicals are bought and sold in kilograms and tons. Because the masses of individual atoms are so minuscule (on the order of 10 −23 g/atom), chemists do not measure the mass of individual atoms or molecules. The problem for Dalton and other early chemists was to discover the quantitative relationship between the number of atoms in a chemical substance and its mass. We also described the law of multiple proportions, which states that the ratios of the masses of elements that form a series of compounds are small whole numbers. In Dalton’s theory each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers. The same calculation can also be done in a tabular format, which is especially helpful for more complex molecules: what is thorium-234 in relion to uranium-238? When was daylight saving first introduce? How many lone pairs does S have in SCl4? What minerals make a oven? Do hairdressers enjoy rubbing against customers? What happens to the shape of an orbit as its eccentricity gets smaller? Morphological expression of a gene? Why is it important to collect forensic evidence and witness accounts as soon as possible after an accident? How does a diagram illustrate newtons third law? Why water is called a universal solvent? Why the diet drink made it float? 15.35 square meters to cubic meters? What is the size of E.\right ) \right ] \) Trending Questions What is a geological column? When was the first plastic ruler made? What describes an observed pattern in nature without attempting to explain it? How do plants like amorseco adapted for reproductive? Were the people in japan prepared for the earthquake 2011? What are the conditions under which consent is said not to be free with example? What is the properties of polymorph? Uranium-238 breaks down into thorium-234.
